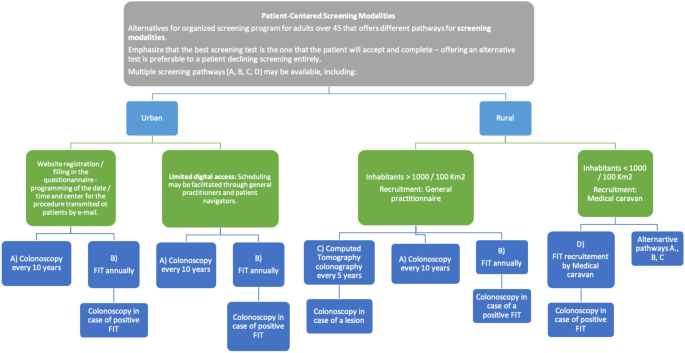
In the current study, we proposed a tailored approach for CRC screening strategies in different areas of the country, correlated with population demographics, disease burden, and regional healthcare system organization.
Economic considerations and screening intervals
The panel’s initial recommendation for annual FIT screening reflects evidence showing superior cost-effectiveness under conditions of high adherence. Recent systematic analyses have indicated that annual FIT screening results in the most significant increase in quality-adjusted life years and is more economically advantageous compared to the absence of screening15,16. However, practical implementation considerations in healthcare systems with limited resources favor biennial screening intervals, as adopted by most European programs17.
Romanian screening strategy published in 2024 by the Romanian Ministry of Health considers biennial FIT as the primary strategy13. However, assuming realistic levels of adherence, conducting annual FIT proved to be the most effective and economical approach16. Cost-effectiveness modeling specific to Romanian healthcare costs and epidemiology will be essential for optimizing the screening interval. Studies from similar healthcare systems suggest biennial FIT remains highly cost-effective, with incremental cost-effectiveness ratios well below accepted thresholds15,17,18. The framework allows flexibility in screening intervals based on regional capacity and performance monitoring.
Comparison with international models
Internationally, there are two models for CRC screening: organized and opportunistic (see supplementary material Table A.3). Organized screening is a dedicated program for CRC screening in which the population is invited for screening, in mass, at a specific age, and follow-up in the long term. Opportunistic screening is used to investigate when patients are admitted for other reasons19. Globally, the most common method for CRC screening is FIT, whereas in the USA and several other regions, including parts of Germany and Poland, colonoscopy is the preferred screening technique19.
Our consensus guidelines emphasize the role of primary care in CRC screening, aligning with findings from both developed and developing healthcare systems that an international Comparison of Programs revealed that 54% of our participants reported no national screening in their country, highlighting the global gap. Regions represented by our respondents with no programs included parts of Eastern Europe, Central Asia, and North Africa. Those from countries with established programs (such as Italy, the UK, Finland, and Belgium) generally rated them as moderately effective. This aligns with reports that even in the EU, many programs have not yet reached high coverage. For instance, one participant from Spain commented that their program was “functional but could be improved: participation approximately 50%”. Participants from countries such as Nepal, Ethiopia, and Algeria pointed out that they rely on opportunistic screening, and that there is much room for improvement.
Analysis of the European Union Member States revealed a wide variability in screening performance, with a participation rate higher for fecal immunochemical testing (FIT) (interval 22.8% – 71.3%) than for guaiac fecal occult blood testing (gFOBT) (4.5 – 67%) (P < 0.001)20. Compliance with colonoscopy varies between 64% and 92%, with a completion rate of 92–99%20.
In our study, we observed specialty differences, with certain opinions varying according to specialty. Surgeons and gastroenterologists, for example, were more confident about colonoscopy (nearly half of the surgeons chose colonoscopy for their own screening). Non-physician participants leaned more towards stool tests. This may reflect knowledge of the test characteristics and personal comfort; providers know the benefits of colonoscopy but are also aware of its burdens, whereas patients favor less invasive options. Importantly, despite these differences, the consensus process led both groups to agree on an FIT-first approach to population screening, recognizing the need to maximize participation.
High-quality studies have consistently demonstrated CRC mortality reduction through screening compared with no screening, notably using gFOBT (relative risk 0.78–0.91), FIT (relative risk 0.90), flexible sigmoidoscopy (incidence risk ratio 0.74), colonoscopy (0.32, 95% CI 0.24–0.45), and CT colonography (sensitivity 0.86–1.0)21.
Our analysis suggests that even among health professionals, their understanding of screening test performance is not uniform. This indicates an opportunity for education as part of implementing a program, not only for the public but also for healthcare providers, so that they can uniformly counsel patients.
The long-term analysis of the UK Flexible Sigmoidoscopy Screening randomized controlled trial showed that once-only flexible sigmoidoscopy was associated with protection for more than 17 years22. The reduction in CRC incidence and mortality was observed to be 26% (hazard ratio [HR] = 0.74, 95% CI 0.70–0.80, P < 0.0001) and 30% (HR = 0.70, 95% CI 0.62–0.79, P < 0.0001), respectively, in the intention-to-treat analysis. In the per-protocol analysis, the reductions were 35% (HR = 0.65, 95% CI 0.59–0.71) and 41% (HR = 0.59, 95% CI 0.49–0.70), respectively22.
A critical benefit of CRC screening programs is the significant shift towards early-stage disease at diagnosis, enabling improved prognosis and treatment outcomes23. Analysis of the Kaiser Permanente Northern California health plan revealed that screening was associated with a decrease in advanced CRCs from 45.9 to 29.3 cases/100000 (P < 0.01) and a 52.4% reduction in mortality from 30.9 to 14.7 deaths/100000 (P < 0.01)23.
CRC screening improves OS and DFS by detecting cancer at an early stage and preventing advanced disease24. An RCT investigated the effect of a single screening colonoscopy performed in healthy individuals aged 55–64 years compared with no screening (11843 versus 56365 patients)25. After a decade, the incidence of colorectal cancer (CRC) was 0.98% in the screening cohort compared to 1.20% in the non-screening cohort, yielding a risk ratio (RR) of 0.82 (95% CI 0.70–0.93). The overall mortality rates were comparable between the screening and non-screening groups, at 11.03% and 11.04%, respectively (RR = 0.99, 95% CI 0.96–1.04). The cancer-specific mortality rates were 0.28% for the screening group and 0.31% for the non-screening group (RR = 0.90, 95% CI 0.64–1.16). To prevent one case of CRC, it was necessary to screen 455 individuals (95% CI 270 – 1429)25. A modified per-protocol analysis of individuals who had a colonoscopy indicated that the likelihood of dying from CRC was reduced by half in the screening group compared to the non-screening group (0.15 versus 0.30, risk ratio – 0.50, 95% CI 0.27–0.77)25.
Implications for healthcare policy
Compared to Western Europe, populations from Eastern, Southern, and Northern Europe were less likely to be updated with CRC screening, with odds ratios (OR) of 0.17, 0.36, and 0.30, respectively26. Less likely to have an undated screening were also people 50–54 years old (OR = 0.59 when compared with 70–74 years old), born outside the EU (OR = 0.85), with low education (OR = 0.61), low income (OR = 0.82), and unemployed (OR = 0.88)26.
In our study, many survey respondents emphasized adherence challenges. In the free text, several noted that “getting people to actually do the test” is the hardest part. The consensus on using the mail-in FIT for rural areas stems from this practical awareness. Additionally, one point raised was the role of GP endorsement; multiple participants cited evidence that a strong recommendation from a physician is a key driver of patient participation. This reinforces our emphasis on actively involving primary care physicians in the program (from risk assessment to recommendation of screening and follow-up to non-responders).
Moreover, colorectal cancer (CRC) screening initiatives are not only highly cost-effective but also have the potential to save money by reducing the financial strain associated with treating advanced cancer. A study conducted in Australia demonstrated that screening is very cost-effective, with expenses per life-year gained amounting to less than $55,000 annually in 2010 Australian currency27. A study conducted in Thailand found that both colonoscopies performed every decade and annual FIT were cost-effective alternatives to no screening. The incremental cost-effectiveness ratios (ICER) for these methods were $600.20 and $509.84 per quality-adjusted life year (QALY) gained, respectively28. Colonoscopy demonstrated greater cost-effectiveness compared to FIT, with an ICER of 646.53 USD per QALY gained28.
Interestingly, some studies have suggested that CRC screening can be cost-effective and cost-efficient. In Vietnam, the total economic burden of CRC was estimated at $132.9 million in 2018, with indirect costs comprising 83.58% of the total cost29. This highlights the potential of screening programs to reduce the economic impact by detecting cancers at earlier stages. Similarly, a Canadian study found that annual FIT screening could save CAN$68 per person over a lifetime compared with no screening30.
Integrated primary care and centralized system approach
The framework integrates primary care physician engagement with centralized program coordination to optimize both accessibility and quality assurance. Primary care physicians serve as the first point of contact for risk assessment, patient education, test distribution, and result communication, leveraging established patient-provider relationships and trust31. Simultaneously, a centralized information system manages population registries, invitation scheduling, result tracking, recall protocols, and quality monitoring consistent with successful European models.
Patient preferences and participation barriers
International studies demonstrate variable patient preferences for colonoscopy versus non-invasive screening methods. Analysis of 1000 participants related to the US Multi-Society Task Force tier 1 tests showed that 68.9% of participants aged 40–49 and 77.4% of people over 50 years old prefer an annual FIT over a colonoscopy each 10 years32. A Korean survey revealed that colonoscopy was preferred in 68.7% of cases over FIT (preferred by 31.3% of respondents)33. Another evaluation from USA showed a slightly preference for FIT over colonoscopy (0.517 versus 0.483)34.
Romanian pilot programs have identified specific barriers including limited health literacy, mistrust of public health initiatives, geographic access challenges in rural areas, and insufficient primary care engagement11,12. Successful implementation requires culturally adapted public education campaigns, streamlined test distribution through primary care, and addressing rural-urban healthcare disparities. Evidence suggests that strong physician recommendation is the most powerful predictor of screening participation, reinforcing the framework’s emphasis on primary care physician engagement. Future Romanian programs should incorporate patient preference assessments and barrier analysis to optimize participation across diverse population segments.
Limitations and future research
Several limitations must be acknowledged. First, the predominance of Romanian professionals (82% in Round 2) may limit international generalizability, though this reflects the study’s focus on developing a Romania-specific framework. Second, the absence of direct patient participation in the consensus process represents a significant limitation; future implementation should incorporate patient preference studies and community engagement. Third, the recommendations lack formal cost-effectiveness analysis specific to Romanian healthcare costs and epidemiology, highlighting the need for economic evaluation studies before national implementation. Fourth, the framework has not undergone pilot testing or validation in Romanian healthcare settings. Finally, the study was conducted during the COVID-19 pandemic, which may have influenced participant perspectives on healthcare system capacity and implementation feasibility. The COVID-19 pandemic disrupted the screening process. Several respondents referenced this: for example, one noted that in their region, “screening colonoscopies were paused for over a year”. Global studies have estimated that pandemic interruptions have led to millions of missed screenings and will likely result in thousands of additional CRC cases and deaths. Our consensus includes planning for catch-up strategies and ensuring the resiliency of screening programs (such as using FIT, which can be performed at home even during pandemics). The survey data showed that participants were acutely aware of these impacts (e.g., 85–95% drops in screening during the peak of the COVID-19 pandemic). On the other hand, the waiting lists for elective diagnostic and therapeutic procedures in symptomatic individuals are increasing in countries around the world, making it challenging to screen asymptomatic patients. An additional challenge for preventive healthcare policies is striking a balance between cancer screening initiatives and addressing other pressing health concerns affecting younger populations such as trauma management and injury prevention.
Despite these limitations, the framework provides evidence-based guidance for Romanian CRC screening program development while acknowledging the need for ongoing refinement based on implementation experience and local data.
The Romanian National Institute of Statistics reported that in 2023 there were 9135391 people over 45 years of age35, translating into an estimation of over 180 centers required for CRC screening. Analyses consistently showed that stool-based tests and colonoscopy-based screening strategies fall below the commonly accepted cost-effectiveness thresholds, making organized CRC screening a sound investment for public health systems. However, successful national implementation will require significant investments in healthcare infrastructure, particularly to address the increased demand for colonoscopy and pathology services following positive screening results.
In conclusion, we propose an algorithm for a structured national CRC screening program, a framework that aligns with the country’s demographics, healthcare resources, and logistical realities while maximizing adherence in the target population.
The current approach proposes a coordinated approach for construction and implementation, considering the robust evidence regarding clear and substantial benefits in reducing CRC mortality, increasing the detection of early-stage cancers, and delivering cost-effective healthcare outcomes.
link