U.S. Gastric Cancer Diagnostics Market Summary
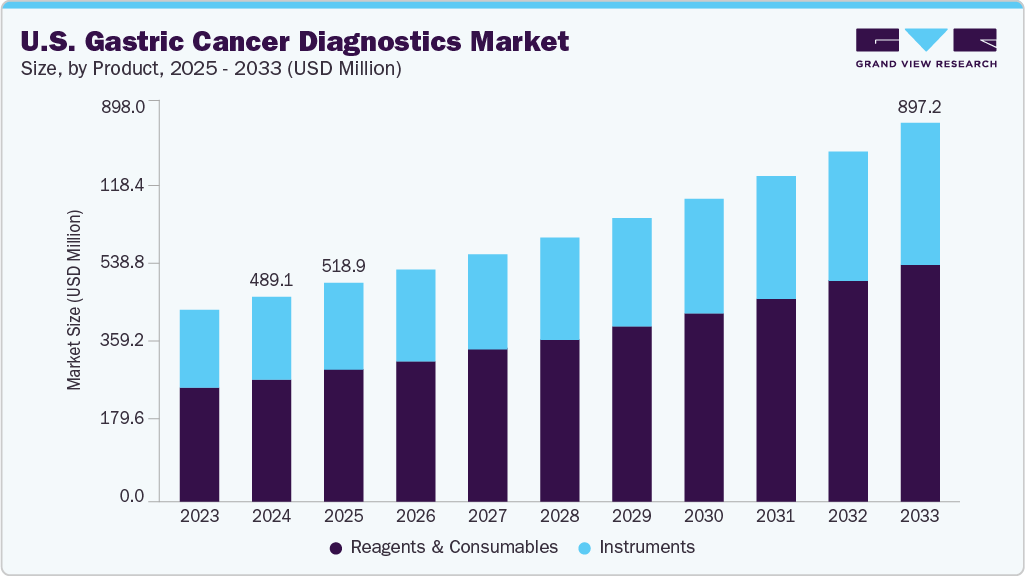
The U.S. gastric cancer diagnostics market size was estimated at USD 489.09 million in 2024 and is projected to reach 897.20 million by 2033, and growing at a CAGR of 7.08% from 2025 to 2033. The U.S. gastric cancer diagnostics market is expanding due to advancements in diagnostic technologies, increasing awareness and patient education, and the expansion of healthcare infrastructure in emerging markets. According to the American Cancer Society (ACS) in 2024, gastric cancer accounted for 26,890 new cases and 10,880 deaths in the U.S. In addition, 30,300 new cases and 10,780 deaths are estimated in the U.S. for the year 2025.
Technological innovations such as liquid biopsies, next-generation sequencing (NGS), and AI-based imaging are propelling the market growth. These tools enable early detection, improved staging, and better characterization of tumor biology, leading to more personalized and effective treatment plans. Advanced endoscopy techniques, like narrow-band imaging and confocal laser endomicroscopy, enhance diagnostic accuracy and patient outcomes. Integration of molecular diagnostics also allows for targeted therapies based on genetic profiles. As healthcare providers and researchers push for more precise, less invasive methods, technological progress boosts market demand, offering faster results, better sensitivity, and reduced patient discomfort compared to traditional methods.
Public awareness campaigns and educational initiatives led by governments, NGOs, and healthcare providers are further fueling market growth by improving recognition of gastric cancer symptoms and risk factors. One such example is Hope for Stomach Cancer, a national nonprofit organization that will play a key role at the May 2025 ASCO Annual Meeting, advancing early detection, treatment, and awareness of stomach and gastroesophageal cancers through advocacy, education, patient support, and national legislative partnerships. This increased awareness is prompting earlier medical consultations and higher participation in screening programs. Moreover, the rise in health literacy and access to online medical information empowers patients to seek timely diagnosis and appropriate care. As patients and caregivers become more proactive, the demand for accurate and accessible diagnostic services grows. This trend is vital in emerging economies, where awareness efforts are narrowing the gap in healthcare utilization and fueling market growth for diagnostic technologies.
The expansion of healthcare infrastructure associated with developing nations is one of the drivers in the U.S. gastric cancer diagnostics market through a different lens, namely, health equity and outreach. In the U.S., ongoing investments in healthcare infrastructure, especially in underserved and rural areas, are improving access to diagnostic services, including endoscopy and advanced imaging for gastric cancer. Federally funded programs and hospital network expansions aim to bridge disparities, enabling earlier detection and better outcomes in high-risk populations such as Asian American and Hispanic communities, where gastric cancer prevalence is comparatively higher. These efforts support market growth by increasing demand for diagnostic tools.
Market Concentration & Characteristics
The U.S. gastric cancer diagnostics market exhibits a high degree of innovation, with advancements in AI-assisted endoscopy, liquid biopsy, and molecular diagnostics. For example, in August 2023, Mirxes Corporation USA, a subsidiary of Mirxes Pte Ltd, announced that its leading product, GASTROClear, a PCR-based in vitro diagnostic test for early gastric cancer detection, received Breakthrough Device Designation from the U.S. Food and Drug Administration (FDA). Ongoing R&D focuses on improving early detection, non-invasive screening, and precision medicine, driving continuous technological evolution and increased adoption of next-generation diagnostic tools for gastric cancer.
Mergers and acquisitions are moderately active in the U.S. gastric cancer diagnostics space, with companies seeking to expand technological capabilities and market share. Acquisitions of AI, digital pathology, and molecular diagnostic firms support integrated solutions, accelerating innovation and enabling broader access to advanced cancer detection technologies.
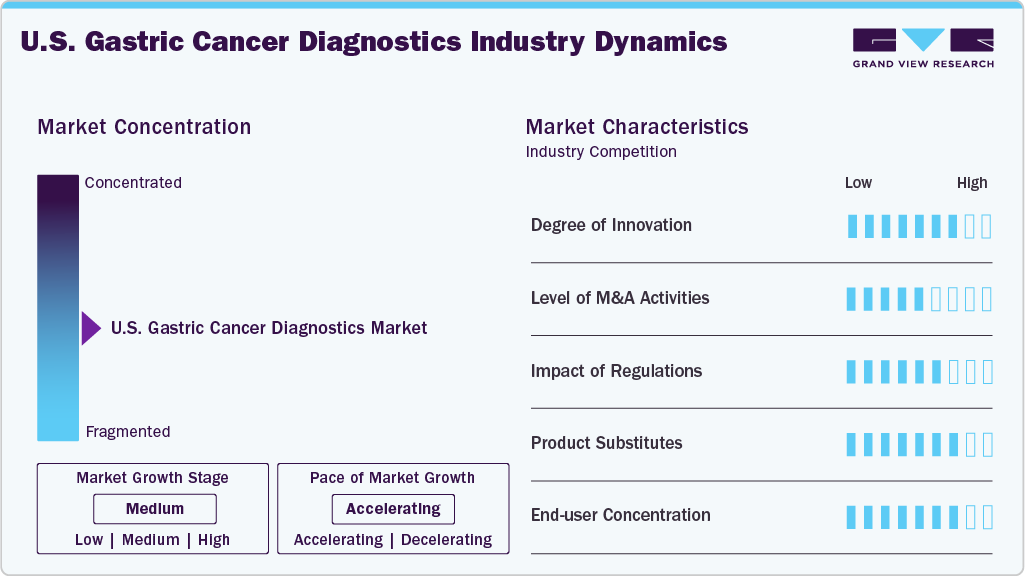
Regulatory frameworks, led by the FDA, play a crucial role in shaping market access and innovation timelines. Strict requirements for clinical validation and approval of new diagnostics ensure safety and efficacy, but can delay market entry. However, fast-track designations for breakthrough technologies can accelerate adoption.
Product substitution remains moderate; although traditional methods like imaging and endoscopy are still used, emerging technologies such as blood-based biomarker tests, liquid biopsy, and AI-assisted diagnostics are gaining ground. For instance, in June 2024, Alverno Laboratories, in partnership with Ibex Medical Analytics, introduced Ibex’s AI platform into routine use across its laboratory network in Illinois and Indiana. Phase 1 of the rollout is complete, featuring AI-powered prostate cancer detection for biopsies, with breast and gastric cancer diagnostics to follow.
The market shows a relatively high end-user concentration, with major demand stemming from large hospitals, cancer centers, and academic institutions. These facilities often drive the adoption of advanced diagnostics, while smaller clinics may lag due to budget or infrastructure constraints, influencing distribution and pricing strategies.
Emerging Diagnostic Technologies Transforming Gastric Cancer Detection and Patient Care
-
Artificial Intelligence (AI)-Enhanced Endoscopy: AI algorithms, including convolutional neural networks (CNNs), analyze endoscopic images to identify gastric cancer with high sensitivity and specificity. Systems like GRAIDS assist clinicians in detecting early gastric cancer, even in challenging cases.
-
Confocal Laser Endomicroscopy (CLE): CLE provides real-time, microscopic imaging during endoscopy, allowing for in vivo optical biopsies. This technique aids in distinguishing malignant from benign tissues, enhancing diagnostic accuracy without the need for traditional biopsies.
-
Liquid Biopsy: Non-invasive blood tests detect circulating tumor DNA (ctDNA), providing insights into genetic mutations and tumor dynamics. Liquid biopsies offer a promising alternative for the early detection and monitoring of gastric cancer.
-
MicroRNA Biosensors: These biosensors detect specific microRNAs associated with gastric cancer. They offer rapid, sensitive, and cost-effective diagnostic options, facilitating early detection through various body fluids.
-
Radiomics: Radiomics involves extracting quantitative features from medical images to develop predictive models. In gastric cancer, it aids in assessing tumor heterogeneity and predicting treatment responses, supporting personalized therapy decisions.
Product Insights
Reagents and consumables dominated the market with a revenue share of 60.88% in 2024 and are projected to grow at the fastest rate in the gastric cancer diagnostics market, driven by their essential role in a wide range of diagnostic procedures, including histopathology, immunohistochemistry, and molecular testing. These products are critical for the preparation, staining, and analysis of tissue and fluid samples, making them indispensable across clinical and research laboratories.
The growing incidence of gastric cancer, combined with the increasing adoption of advanced diagnostics such as biomarker-based testing and next-generation sequencing, is fueling demand for high-quality, specific reagents and assay kits. Furthermore, as testing volumes rise due to early detection programs and personalized medicine approaches, the need for frequent replenishment of consumables boosts recurring revenue, making this segment highly lucrative. The expansion of automated platforms and digital pathology further increases reagent consumption, as standardized protocols require compatible, ready-to-use kits, ensuring consistent results and improved diagnostic accuracy across healthcare settings.
Disease Type Insights
Adenocarcinoma is the largest segment with a revenue share of 85.64% in 2024 and is expected to witness the fastest CAGR over the forecast period, accounting for the majority of gastric cancer cases globally. According to an American Cancer Society (ACS) article published in April 2024, adenocarcinoma accounts for two-thirds of esophageal cancer cases in high-HDI (human development index) countries, linked to obesity, GERD, and Barrett’s esophagus.
Rising incidence rates suggest that increasing excess body weight will significantly contribute to the future burden of this cancer type. Originating in the glandular cells of the stomach lining, adenocarcinoma is the most prevalent histological subtype. It is often associated with risk factors such as Helicobacter pylori infection, smoking, dietary habits, and chronic gastritis. Its dominance in clinical incidence makes it a primary focus for diagnostic efforts, driving demand for precise and early detection methods.
Diagnostic tools, including endoscopy, biopsy, immunohistochemistry, and molecular profiling, are widely employed to confirm and characterize adenocarcinoma. The push toward early diagnosis and personalized therapy further accelerates the need for biomarker-based tests specific to this subtype. In addition, advancements in genomic sequencing and liquid biopsy are increasingly being integrated into adenocarcinoma detection strategies. The rising global burden of this cancer type ensures its continued significance within the diagnostic landscape, fueling growth in this segment.
End-use Insights
Hospitals represented the largest segment with a revenue share of 64.96% in 2024 in the U.S. gastric cancer diagnostics market. This growth is driven by the increasing adoption of advanced diagnostic technologies, such as artificial intelligence (AI) and machine learning (ML), which enhance diagnostic precision and efficiency. Hospitals offer comprehensive services, including initial screenings, advanced diagnostics, and treatment, making them preferred centers for gastric cancer care. The integration of AI and ML into diagnostic procedures further boosts the demand for hospital-based services, contributing to the segment’s rapid expansion.
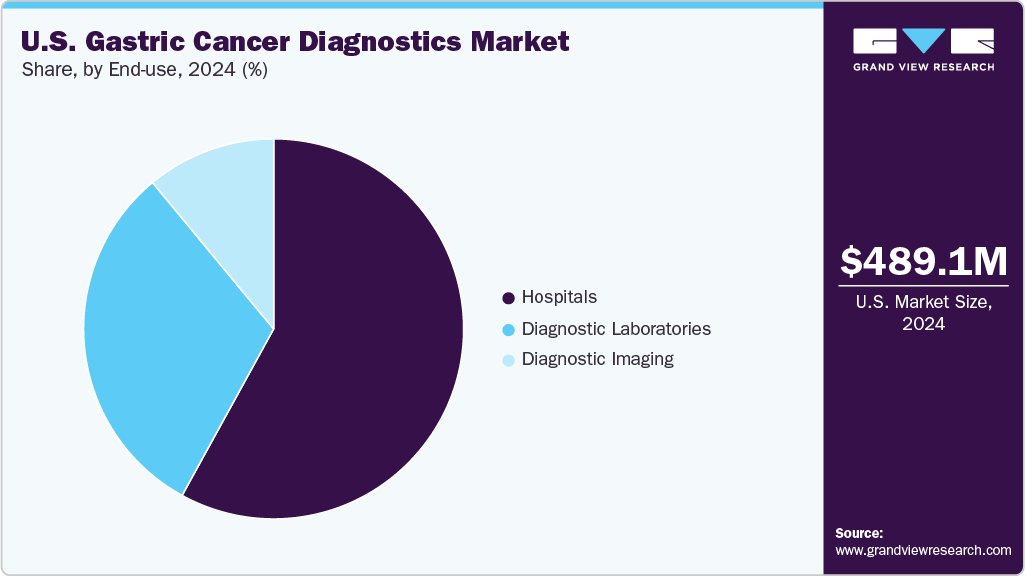
Diagnostic laboratories represent the fastest-growing segment due to their central role in testing, analyzing, and confirming gastric cancer cases. These laboratories perform a wide range of diagnostic procedures, including histopathology, molecular testing, immunohistochemistry, and biomarker analysis, essential for accurate diagnosis and personalized treatment planning. In addition, the integration of technologies such as next-generation sequencing (NGS), liquid biopsy, and AI-assisted pathology has enhanced the capabilities of diagnostic labs, further boosting their market share. For example, in March 2025, Paige launched the Paige GI Suite. This AI-driven diagnostic tool aids pathologists in analyzing biopsies across the entire gastrointestinal tract, including the esophagus, stomach, duodenum, and colorectum. Moreover, centralized diagnostic facilities provide faster turnaround times and standardized results, improving clinical decision-making and patient outcomes. These factors collectively establish diagnostic laboratories as the dominant segment within the gastric cancer diagnostics market.
Country Insights
The U. S. holds a significant revenue share in the global gastric cancer diagnostics market, driven by advanced healthcare infrastructure, high healthcare expenditure, and a growing focus on early cancer detection. Despite gastric cancer being less prevalent in the U.S. compared to some Asian countries, increasing awareness and adoption of advanced diagnostic technologies contribute to market growth. The availability of state-of-the-art diagnostic tools such as endoscopy, molecular testing, and AI-powered pathology solutions supports precise and early diagnosis, improving patient outcomes.
Government initiatives and private sector investments aimed at cancer screening and precision medicine also stimulate market expansion. The presence of leading diagnostic companies and research institutions fosters innovation and continuous introduction of novel biomarkers and minimally invasive tests, positioning the U.S. as a key market for gastric cancer diagnostics.
Key U.S. Gastric Cancer Diagnostics Company Insights
Key players in the U.S. gastric cancer diagnostics market include Natera, Inc., Illumina, GE Healthcare, QIAGEN, and Thermo Fisher Scientific. The adoption of cutting-edge technologies such as artificial intelligence, next-generation sequencing, and liquid biopsy is greatly enhancing the accuracy and efficiency of cancer diagnostics. These innovations reflect the market’s ongoing evolution, with both major companies and emerging technologies contributing to its dynamic growth and advancement.
Key U.S. Gastric Cancer Diagnostics Companies:
- Illumina, Inc
- Guardant Health
- Natera, Inc
- Thermo Fisher Scientific
- QIAGEN
- Freenome Holdings Inc.
- Exact Sciences Corporation
- PathAI, Inc
- GE Healthcare
Recent Developments
-
In January 2025, Geneoscopy secured USD 105 million in Series C funding, led by Bio-Rad, to advance its next-generation gastrointestinal diagnostic technologies and support the upcoming launch of its noninvasive colorectal cancer test, ColoSense.
-
In June 2024, Alverno Laboratories, in partnership with Ibex Medical Analytics, introduced Ibex’s AI platform into routine use across its laboratory network in Illinois and Indiana. Phase 1 of the rollout was completed, featuring AI-powered prostate cancer detection for biopsies, with breast and gastric cancer diagnostics to follow.
-
In August 2023, Mirxes Corporation USA, a subsidiary of Mirxes Pte Ltd, announced that its leading product, GASTROClear, PCR-based in vitro diagnostic test designed for the early detection of gastric cancer received Breakthrough Device Designation from the U.S. Food and Drug Administration (FDA).
U.S. Gastric Cancer Diagnostics Market Report Scope
|
Report Attribute
|
Details
|
|
Market size value in 2025
|
USD 518.94 million
|
|
Revenue forecast in 2033
|
USD 897.20 million
|
|
Growth rate
|
CAGR of 7.08% from 2025 to 2033
|
|
Actual data
|
2021 – 2024
|
|
Forecast period
|
2025 – 2033
|
|
Quantitative units
|
Revenue in USD million/billion and CAGR from 2025 to 2033
|
|
Report coverage
|
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
|
|
Segments covered
|
Product, disease type, end-use
|
|
Country scope
|
U.S.
|
|
Key companies profiled
|
Illumina, Inc.; Guardant Health, Natera, Inc; Thermo Fisher Scientific; QIAGEN; Freenome Holdings Inc; Exact Sciences Corporation; PathAI, Inc; GE Healthcare
|
|
Customization scope
|
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, & segment scope
|
|
Pricing and purchase options
|
Avail customized purchase options to meet your exact research needs. Explore purchase options
|
U.S. Gastric Cancer Diagnostics Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. gastric cancer diagnostics market report based on product, disease type, end use:
-
Product Outlook (Revenue, USD Million, 2021 – 2033)
-
Reagents & Consumables
-
Instruments
-
-
Disease Type Outlook (Revenue, USD Million, 2021 – 2033)
-
Adenocarcinoma
-
Gastric lymphoma
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 – 2033)
-
Hospitals
-
Diagnostic Laboratories
-
Diagnostic imaging
-
Frequently Asked Questions About This Report
b. The U.S. gastric cancer diagnostics market size was estimated at USD 489.09 million in 2024 and is expected to reach USD 518.94
million in 2025.
b. The U.S. gastric cancer diagnostics market is expected to grow at a compound annual growth rate of 7.08% from 2025 to 2033 to reach USD 897.20 million by 2033.
b. Reagents and consumables dominate the market with a revenue share of 60.88% in 2024 and is the fastest-growing segment in the gastric cancer diagnostics market, driven by their essential role in a wide range of diagnostic procedures, including histopathology, immunohistochemistry and molecular testing. These products are critical for the preparation, staining, and analysis of tissue and fluid samples, making them indispensable across clinical and research laboratories.
b. Some key players operating in the U.S. gastric cancer diagnostics market include Illumina, Inc, Guardant Health, Natera, Inc, Thermo Fisher Scientific, QIAGEN, Freenome Holdings Inc, Exact Sciences Corporation, PathAI, Inc, GE Healthcare
b. The U.S. gastric cancer diagnostics market is expanding due to advancements in diagnostic technologies, increasing awareness and patient education and expansion of healthcare infrastructure in emerging markets. According to the American cancer society in 2024, gastric cancer accounted for 26,890 new cases and 10,880 deaths in U.S. In addition, 30,300 new cases and 10,780 deaths are estimated in the U.S. for the year 2025.
link
